
-
 03/172026BIOREGENIX's Collagen Fiber Filler-III Registration Application Accepted by NMPA
03/172026BIOREGENIX's Collagen Fiber Filler-III Registration Application Accepted by NMPA -
 03/0220262026 BIOREGENIX REVICARA Academic Exchange Conference Successfully Concludes
03/0220262026 BIOREGENIX REVICARA Academic Exchange Conference Successfully Concludes -
 02/052026Bairen Medical BIOREGENIX Industrial Base Project Selected as One of Beijing's "3×100" Key Projects for 2026
02/052026Bairen Medical BIOREGENIX Industrial Base Project Selected as One of Beijing's "3×100" Key Projects for 2026 -
 12/272025December Highlights | Winning Industry Awards, Shaping a New Future Together
12/272025December Highlights | Winning Industry Awards, Shaping a New Future Together -
 12/122025China's First Cheek-Specific Collagen Implant Launched
12/122025China's First Cheek-Specific Collagen Implant Launched -
 09/142025At CIFTIS 2025, BIOREGENIX Brings Chinese Quality to the Global Stage
09/142025At CIFTIS 2025, BIOREGENIX Brings Chinese Quality to the Global Stage -
 08/312025August Highlights丨Riding the Wave of Industry Events, Driving Innovation
08/312025August Highlights丨Riding the Wave of Industry Events, Driving Innovation -
 08/112025BIOREGENIX's Filler-II Registration Application Accepted by NMPA
08/112025BIOREGENIX's Filler-II Registration Application Accepted by NMPA -
 07/192025BIOREGENIX Team Building in Huairou
07/192025BIOREGENIX Team Building in Huairou -
 06/202025BIOREGENIX Attends 2025 Collagen Multi-field Forum
06/202025BIOREGENIX Attends 2025 Collagen Multi-field Forum -
 05/212025BIOREGENIX Named Among Top 20 Pioneer Enterprises in Boundary-Breaking Innovation
05/212025BIOREGENIX Named Among Top 20 Pioneer Enterprises in Boundary-Breaking Innovation -
 04/042025BIOREGENIX Showcases at BIA 2025 Asia Injection Medical Aesthetics Summit
04/042025BIOREGENIX Showcases at BIA 2025 Asia Injection Medical Aesthetics Summit -
 12/072024Aikeyan Launches Official Account on Xiaohongshu
12/072024Aikeyan Launches Official Account on Xiaohongshu -
 11/242024BIOREGENIX Debuts at Beijing Life Science Forum 2024
11/242024BIOREGENIX Debuts at Beijing Life Science Forum 2024 -
 10/292024BIOREGENIX's Filler-I Registration Application Accepted by NMPA
10/292024BIOREGENIX's Filler-I Registration Application Accepted by NMPA -
 10/2820242024 China Medical Aesthetics Industry "Innovative Potential Enterprise Award"
10/2820242024 China Medical Aesthetics Industry "Innovative Potential Enterprise Award" -
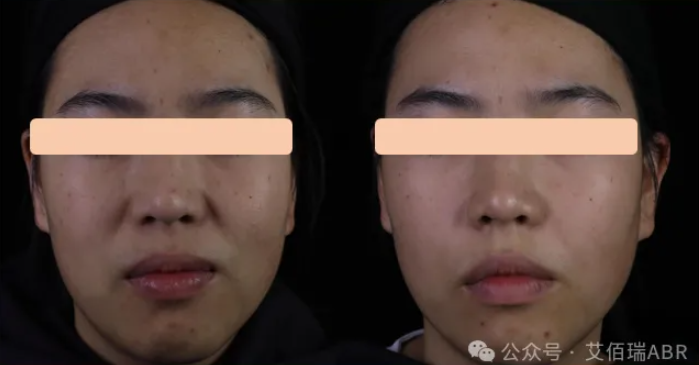 05/162024Collagen Filler-III Wraps Up Clinical Trial Enrollment
05/162024Collagen Filler-III Wraps Up Clinical Trial Enrollment -
 01/192024Baishuang® Wraps Up Clinical Trial Enrollment
01/192024Baishuang® Wraps Up Clinical Trial Enrollment -
 12/182023Innovative Cross-linked Collagen Dermal Filler Successfully Initiates Enrollment
12/182023Innovative Cross-linked Collagen Dermal Filler Successfully Initiates Enrollment -
 09/282023First Collagen Skin Booster Completes Trial Enrollment
09/282023First Collagen Skin Booster Completes Trial Enrollment

